|
8/24/2023 0 Comments Iodine clock reaction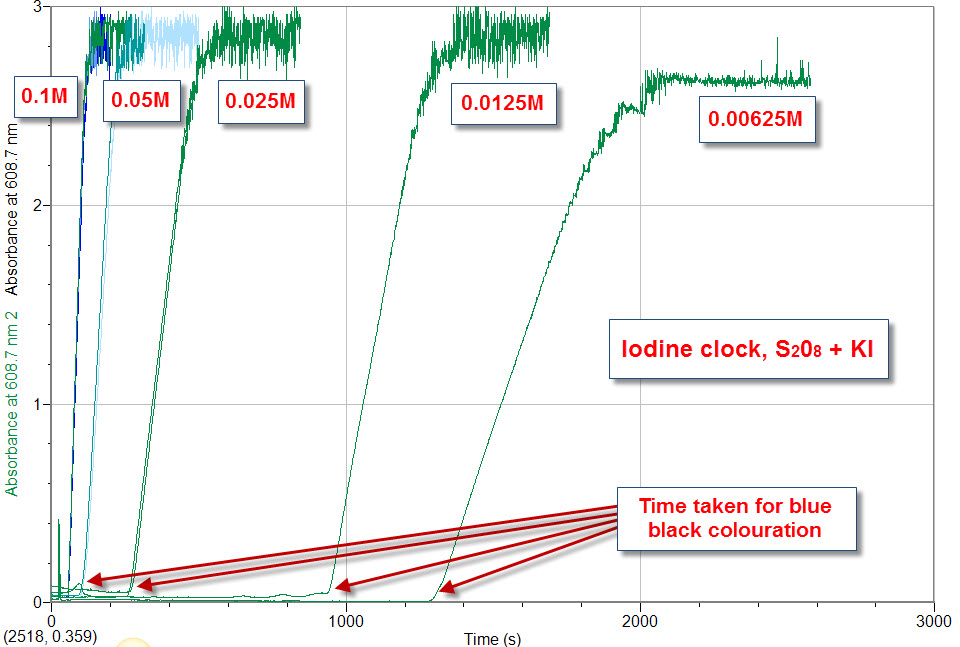
In the iodine clock reaction, the overall reaction process is described by the following mechanism This reaction is a well known example of the so-called "clock reactions" where a reaction displays a clear "endpoint" that appears after a well-defined amount of time. Repeat reaction for solutions thermally equilibrated in ice water, and heated to near boiling. Add solution of potassium iodate to sodium sulfate and starch solution.Ħ. Prepare solutions of starch and combine with solution of sodium thiosulfate.Ĥ. Prepare solutions of sodium thiosulfate.Ģ. Ingredients: sodium bisulfite, potassium iodate, starchġ. 
The iodine "clock" reaction is run at a three temperatures to observe the dependence of the reaction time on the temperature of the reaction solution. the time from mixing equal volumes of the two solution until the first appearance of the blue colour of the starch-iodide complex).Ģ vol H 2O 2 (0.6% w/v) in 0.2 mol dm -3 H 2SO 4Ġ.5% soluble starch solution containing 0.001 mol dm -3 KI and 0.Exploring the temperature dependence of reaction rates using the Landolt "iodine clock" reaction The following concentrations at room temperature gave a time of about 40 seconds for the 'clock' (i.e. Similar results can be obtained by varying the concentration of hydrogen peroxide, concentration of acid or by varying the temperature. Vary the concentration of potassium iodideīoth the rate of reaction as given by the slopes of the lines and the time taken for the appearance of the blue starch-iodide complex depend on the iodide concentration. The time taken for the appearance of the blue starch-iodide complex depends on the thiosulphate concentration. The rate of reaction as given by the slopes of the lines is unchanged. The reaction was performed at room temperature using the reaction mixture described in ‘Methods’. Typical set of results using continuous colorimetry to give accurate determinations of both the time to the appearance of the blue starch-iodide complex and the rate of the reaction (slope of the line). Than 18 'vol' (5% w/v, 1.5 mol dm -3) but weaker than 28 'vol' should Hydrogen peroxide solutions equal to or stronger 3 but weaker than 1.5 mol dm -3 should be labelled Sulphuric acid solutions equal to or stronger than 0.5 mol dm The experiments described here do not useĬoncentrations of reagents that pose a hazard or require warning labels. Potassium iodide, concentration of hydrogen peroxide and concentration of  
The rate of the reaction will depend on temperature, concentration of In fact the kinetics of this reaction can be found without the need for theĪddition of sodium thiosulphate since its only purpose is to delay the appearance Irrespective of the concentration of thiosulphate. Reaction can be obtained from the slope of the line of absorbance against time, Reaction rate, hence the name 'clock reaction'. Taken for the blue colour to first appear is usually used as the indicator of the With a fixed concentration of thiosulphate the time The time taken for the blue colour of the starch-iodide complex to startĪppearing depends on the rate of the initial reaction and on the concentration of The blue complex does not start to form until all the thiosulphate has been However the presence of thiosulphate rapidly converts the triiodide to iodide so Will react with starch producing the characteristic blue starch-iodide complex. When these are mixed triiodide ions are slowly produced. A number ofĭifferent clock reactions have been described, some of which are variations ofįor the clock reaction described here one solution contains hydrogen peroxideĪnd sulphuric acid, the other contains potassium iodide, sodium thiosulphate and Iodine clock was first described by Hans Heinrich Landolt in 1886. Mixing substances which, after a delay, suddenly start to change colour. Clock reactions investigate reaction kinetics by
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
